Three ITS Students Utilize Byproduct of Biodiesel Synthesis

The ITS Student Team that won a gold medal at the 33rd Pimnas in the category of Penelitian Eksakta
ITS Campus, ITS News – Production of used cooking oil caused by the increased consumption of cooking oil in the community is estimated to reach twenty thousand tons annually. Some people have used this used cooking oil as biodiesel, but crude glycerol which is a by-product is still wasted. Concerned by this problem, three students from the Department of Chemistry, Institut Teknologi Sepuluh Nopember (ITS) gave birth to an innovation to convert this waste into a useful lactic acid.
Gardini Nilasari as the coordinator of the team revealed that this research initially referred to the accumulation of crude glycerol in the Laboratory of Instrumentation and Analytical Science at the ITS Chemistry Department. This product is a by-product of the used cooking oil transesterification process in biodiesel synthesis. “If it is thrown away it will cause new waste, even though the original purpose of utilizing used cooking oil is to reduce waste,” she explained.
Crude glycerol itself has a low level of purity and economic value, so it needs to be utilized by converting it into several products. These products include formic acid, oxalic acid, glyceric acid, 1,2-propanadiol, and lactic acid. In the research conducted by Gardini and her two team members, Annisa Putri Agustin and Dewi Septiningtyas, in the Program Kreativitas Mahasiswa (PKM), the by-product was converted into lactic acid.
Why lactic acid is selected as the conversion product, according to Gardini, is due to its massive use in industry. This makes the demand for lactic acid increase and is predicted to reach 1.96 million tons in 2025. “Lactic acid also has the potential to be used as a plastic material for Poly Lactic Acid (PLA) which is easily biodegradable, thus supporting the Sustainability Development Goals (SDGs), of which purpose is to protect marine and terrestrial ecosystems,” she explained again.
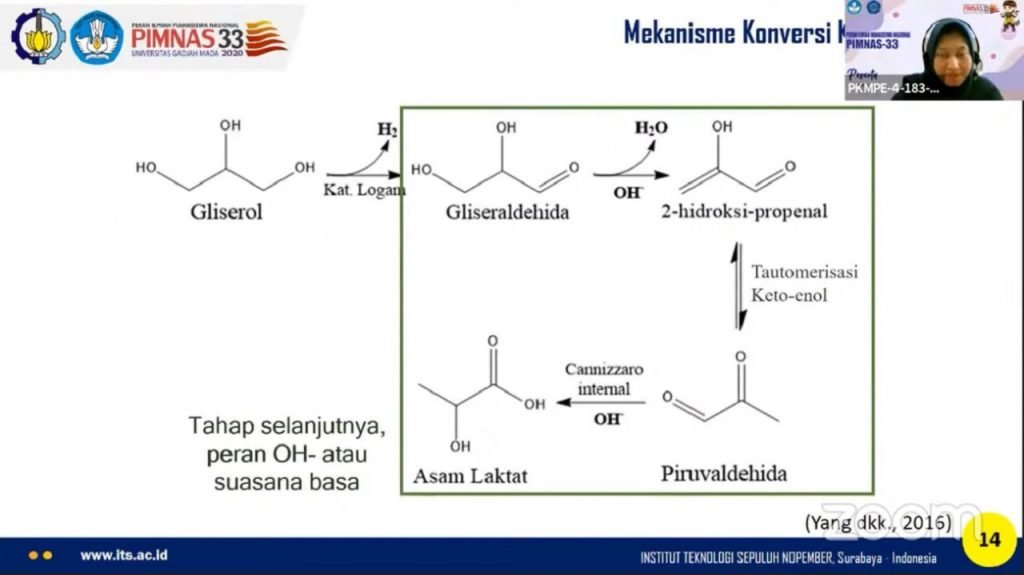
Regarding the conversion process, Gardini and his two colleagues used a hydrothermal catalytic conversion method with a Ni / graphite catalyst. The method was chosen with the hypothesis that high lactic acid yields would be obtained compared to conventional methods (fermentation). Also, there is no need for a purification process for crude glycerol so it is more efficient and the time required for the reaction is relatively fast.
The production of lactic acid using a catalyst (a substance to accelerate the reaction) of Ni / graphite nanoparticles has a high conversion rate and selectivity, because Ni / graphite is nano-sized and has a large surface area. So that it supports the dehydrogenation process (releasing hydrogen atoms, red) glycerol to become glyceraldehyde. In addition, the catalyst does not easily reduce its activity and selectivity.
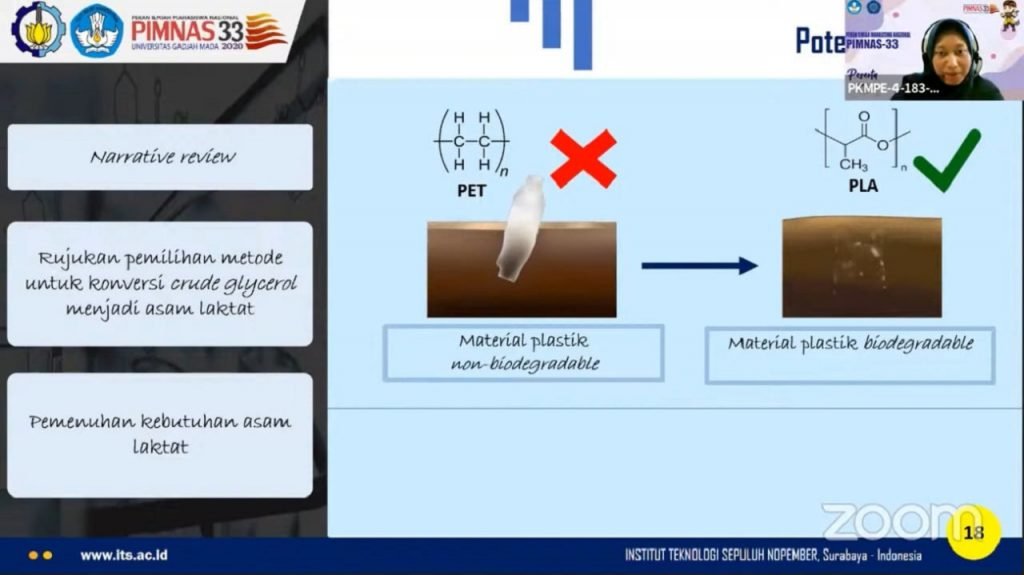
The explanation of the benefits of Lactic Acid as an easily biodegradable PLA plastic material
In this method, it is necessary to add sodium hydroxide to create an alkaline atmosphere. Crude glycerol itself already has a degree of acidity ranging from 8.93 to 9.4 which is classified as a base, so the problem in this method can be resolved. “As additional information, the high degree of acidity is obtained from the residual base catalyst in the transesterification process,” said Gardini.
The Covid-19 pandemic led to data transformation and output targets in the research of the three students under the guidance of Dr. Hendro Juwono MSi. Initially, the discussion related to the composition of crude glycerol used cooking oil, the results of the catalytic conversion of glycerol to lactic acid, and the ratio of glycerol and sodium hydroxide will be discussed based on primary data and laboratory tests. However, with the transformation happening, the research changed to using secondary data from the literature study for approximately two and a half months.
Gardini admitted that her team faced many challenges in the literature study process, where each literature must have high credibility and also can strengthen every argument. “We also have to come up with updates and strengthen the creativity of our topic in writing the output targets of this PKM, such as narrative reviews, progress reports, and final reports,” she explained.

(from left) The ITS student team composed by Annisa Putri Agustin, Gardini Nilasari, Dewi Septiningtyas H
With the creativity they brought, Gardini and her team had the opportunity to become a part of the ITS contingent at the 33rd Pimnas and won gold in the PKM presentation of Penelitian Eksakta category, some time ago. “We are very happy to make ITS proud at the end of our studying period at the kampus perjuangan,” she said emotionally.
Gardini hopes that in the future there will be students who are interested in this topic and can carry out tests in the laboratory, especially those related to optimization of reaction parameters or other supportive matters. “So that in corresponding with the development plan that we describe, the production of lactic acid and the application of its use can become PLA on an industrial scale,” she said positively. (tri / nadh / ITS Public Relations)
Related News
-
Supporting the Development of Material Processes, ITS Professors Utilize Biomass Waste
ITS Campus, ITS News — Innovation in material processes continues to develop to support society’s need for environmentally friendly
January 05, 2021 17:01 -
ITS Professor Ideas for Bio-Corrosion Control in Marine Structures
ITS Campus, ITS News — Coastal and offshore buildings interacting directly with seawater trigger damage, including bio-corrosion. If not
January 05, 2021 17:01 -
ITS Launches the First Marine Floating Solar Power Plant Prototype in Indonesia
ITS Campus, ITS News — Institut Teknologi Sepuluh Nopember (ITS)‘s commitment in realizing the energy transition is getting serious.
January 05, 2021 17:01 -
Targeting Champion, Bayucaraka ITS Launches Flying Robot Innovation
ITS Campus, ITS News – The Bayucaraka team at Institut Teknologi Sepuluh Nopember (ITS) has launched four new flying
January 05, 2021 17:01
